Abstract
The results of clinical trials evaluating CD19-targeting chimeric antigen receptor (CAR) T cells are impressive, with overall response rates of up to 90% in B cell acute lymphoblastic leukemia (ALL) and 50-80% in lymphoma. Despite initial responses, antigen-negative relapse is common following treatment with CD19-targeted therapies and is estimated to occur in up to 39% of patients. One approach of addressing this problem is to utilize dual-targeting CAR T cells, a strategy that has recently been applied to CD19/CD22 for ALL, CS1/BCMA, and BCMA/GPRC5D for multiple myeloma. Dual-targeting CARs can simultaneously target two tumor antigens and, therefore, potentially eradicate heterogeneous tumors. The initial response to dual-targeted CAR T cells is expected to provide greater tumor coverage compared to single-targeted therapy, and can potentially circumvent antigen escape. Moreover, if one tumor antigen becomes downregulated during treatment, the second targeting domain will continue to be reactive to tumors. Therefore, it is critical to identify novel targets that can be combined with CD19 in a dual-targeted immunotherapeutic platform.
To expand the potential for dual targeting of ALL, we developed a CAR T cell therapy against a novel target, B-cell activating factor receptor (BAFF-R), based on the remarkable specificity of anti-BAFF-R antibodies that we previously generated. BAFF-R is expressed almost exclusively on B cells, including in patients with CD19-negative relapse, making it an ideal immunotherapeutic target. Studies demonstrate that the role of BAFF-R in B cell function and survival is conclusive, an important feature that may mitigate the tumor's ability to escape therapy through antigen loss, particularly if a non-redundant role for BAFF-R is confirmed. BAFF-R-CAR T cells demonstrate in vitro effector function and in vivo therapeutic efficacy in CD19-negative models, including patient-derived xenograft models, and are currently being evaluated clinically for the treatment of ALL (NCT04690595). We hypothesized that simultaneous targeting of CD19 and BAFF-R in a bispecific CAR platform could confer a therapeutic advantage and avoid the challenges of sequential administration of CD19 and BAFF-R monospecific CAR T cells.
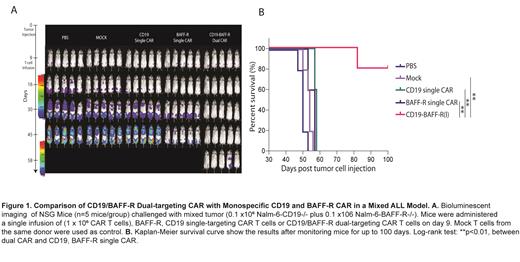
We leveraged our experience with CD19- and BAFF-R-CAR T cells to develop a dual-targeting, bispecific CAR with a 41BB costimulatory domain (CD19/BAFF-R dual CAR). Here, we identified the optimal orientation of the single-chain variable fragment (loop scFv) domains within the dual construct and tested the CD19/BAFF-R dual CAR T cells for their in vitro effector function and in vivo anti-leukemia activity. To evaluate the specific targeting to CD19 and BAFFR, we developed Nalm-6-BAFF-R-knockout (KO) and Nalm-6-CD19-KO cell lines. CD19/BAFF-R dual CAR T cells specifically released IFN-g following incubation with Nalm-6 CD19-/- or BAFF-R-/- cells (P<0.001) compared with un-transduced mock T cells. Both CD4+ and CD8+ CAR T cell populations exhibited effector function. To evaluate the antigen-dependent targeting of the CD19/BAFF-R dual CAR T cells in vivo, we utilized a mixed B-cell leukemia model that simulates clinical tumor heterogeneity. NOD-scid IL2Rgammanull (NSG) mice were inoculated with 2-3x10 5 of mixed Nalm-6 BAFF-R-/- and CD19-/- cells at a 1:1 ratio with a single injection. 1x10 6 CD19/BAFF-R dual CAR, CD19, or BAFF-R single CAR-T cells were administered intravenously 9-10 days later. Tumor growth was monitored by bioluminescent imaging weekly. We observed superior tumor eradication (P<0.01) and survival (P<0.01) (Figure 1) by CD19/BAFF-R dual CAR T cells compared to either single-targeting CAR and mock T cells. The adoptively transferred CD19/BAFF-R dual CAR T cells were able to persist in vivo.
Our unique CD19/BAFF-R dual-targeting CAR T cells will be the first to target this combination of tumor-associated antigens. Our study demonstrated the reliability of bispecific CD19/BAFF-R dual CAR T cell therapy in inducing remission in ALL consisting of CD19-/- and BAFF-R-/- tumors. We hypothesize that simultaneous immunotherapy targeting of heterogeneous leukemic cell populations may diminish the likelihood of antigen escape and may have a significant impact on leukemia treatment by improving the therapeutic benefits of CAR T cell therapy.
Wang: Pepromene Bio, Inc.: Consultancy. Forman: Mustang Bio: Consultancy, Current holder of individual stocks in a privately-held company; Allogene: Consultancy; Lixte Biotechnology: Consultancy, Current holder of individual stocks in a privately-held company. Kwak: Pepromene Bio, Inc.: Consultancy, Current equity holder in publicly-traded company. Qin: Pepromene Bio, Inc.: Consultancy, Current equity holder in publicly-traded company.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal